Key takeaways
- AI assistants like ChatGPT, Perplexity, and Google AI Overviews are now a primary research channel for patients and physicians -- what they say about your brand matters as much as your own website.
- Healthcare and pharma face unique compliance risks when AI models surface inaccurate drug information, outdated clinical claims, or off-label content.
- Trust signals (E-E-A-T, verified reviews, clinical credentials) directly influence whether AI models cite your brand positively or at all.
- Monitoring alone is not enough -- you need to close the loop by identifying content gaps and publishing material that AI models can actually cite.
- Tools like Promptwatch are built for exactly this workflow: track what AI says, find what's missing, create content that gets cited.
Why AI search is a patient safety issue, not just a marketing one
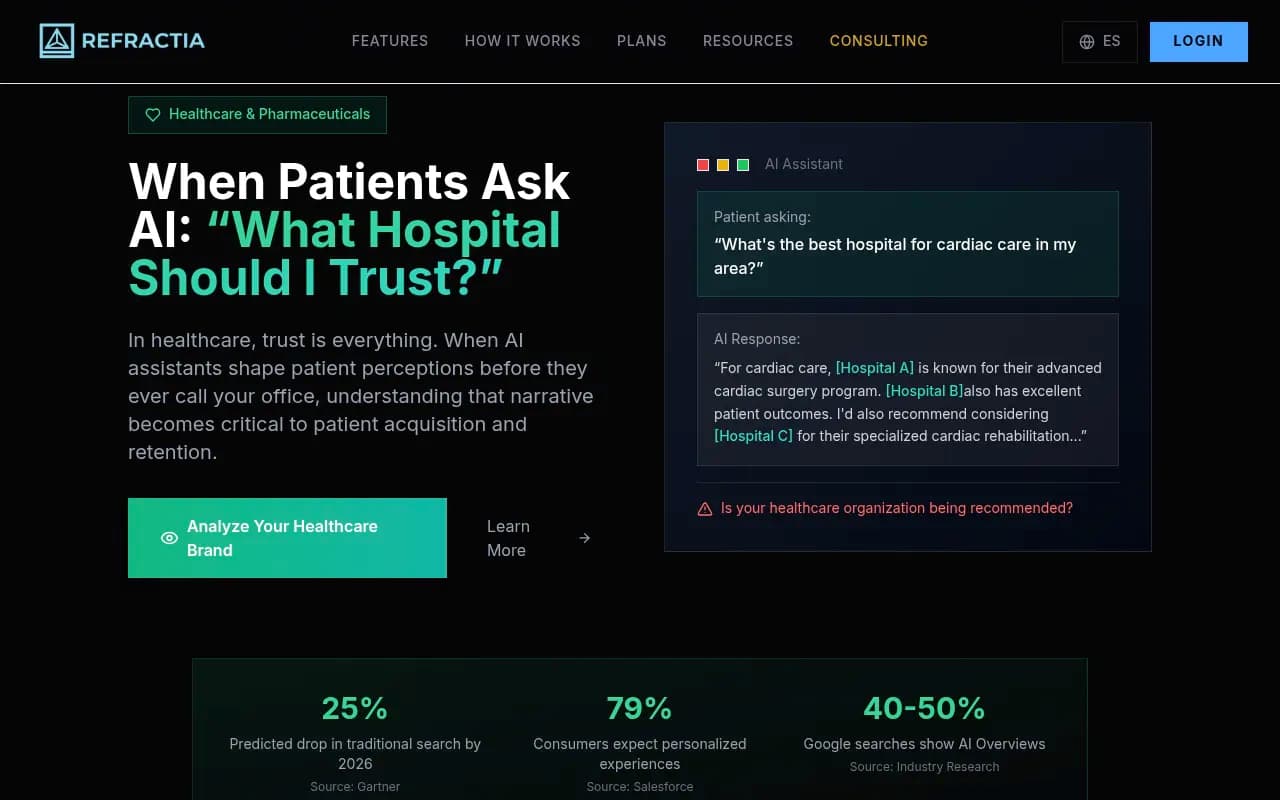
When a patient types "best treatment centers for early-stage breast cancer near me" into ChatGPT or Perplexity, they are not browsing a list of blue links. They are reading a synthesized recommendation. That recommendation either includes your hospital or it doesn't. And if it does include you, it either describes your capabilities accurately or it doesn't.
That second scenario is the one most healthcare marketers haven't fully reckoned with yet.
According to Gartner, traditional search volume is expected to drop roughly 25% by 2026 as AI-generated answers absorb more queries. Google AI Overviews already appear on 40-50% of searches. For healthcare queries -- which tend to be high-stakes, question-based, and research-heavy -- the shift toward AI-mediated answers is happening faster than in most other verticals.
The problem is that AI models pull from a wide and sometimes outdated mix of sources: your website, medical journals, patient review platforms, Reddit threads, news articles, and more. If your oncology program expanded two years ago but your website content still reflects the old scope, an AI model might describe you as a "general hospital" while your competitor's updated content gets them cited as the regional specialist.
That's a lost patient. In pharma, it could be worse.
The compliance dimension pharma brands can't ignore
For pharmaceutical companies, the stakes around AI-generated content go beyond reputation. They touch regulatory compliance directly.
AI models can and do describe drugs in ways that are technically inaccurate -- citing outdated efficacy data, misrepresenting approved indications, or drawing comparisons to competitor products in ways that wouldn't pass FDA or EMA review if a human marketer wrote them. The difference is that no human marketer wrote them. An AI model synthesized them from whatever sources it had access to.
This creates a genuinely new compliance problem. Your regulatory team can review your promotional materials. They can't review every ChatGPT response that mentions your drug.
What they can do is:
- Monitor AI outputs systematically to catch inaccurate or off-label representations
- Ensure your owned content is authoritative enough that AI models prefer it as a source
- Flag and document instances where AI-generated content may constitute a compliance risk
- Work with medical affairs to publish accurate, citable content that displaces bad sources
The February 2026 regulatory update from aihealthcarecompliance.com notes that healthcare AI systems are now considered "high-impact" under emerging consultation frameworks, with patient safety implications driving stricter scrutiny. That scrutiny is aimed at AI tools used in clinical settings, but the downstream effect on how brands manage AI-generated content about their products is real.
Pharmacovigilance teams are already using AI to scan literature for adverse event signals -- over 2.5 million scientific articles are published annually, and AI filters are now capable of screening out 55% of irrelevant articles while retaining 99% of suspected adverse-event reports (per IntuitionLabs' analysis of recent proof-of-concept work). The same principle applies to brand monitoring: you need automated systems watching what AI says about your products, not manual spot-checks.
What trust signals actually influence AI citations in healthcare
AI models don't cite brands randomly. They apply something close to Google's E-E-A-T framework (Experience, Expertise, Authoritativeness, Trustworthiness) when deciding which sources to surface. In healthcare, this plays out in specific ways.
Clinical credentials and author expertise
Content authored or reviewed by named physicians, with credentials clearly stated, is more likely to be cited than anonymous marketing copy. If your blog posts don't have a medical reviewer byline, that's a gap worth fixing. AI models can read schema markup and structured data -- author credentials embedded in your content's metadata signal legitimacy.
Verified reviews and patient satisfaction data
Patient reviews on Google, Healthgrades, and similar platforms feed directly into AI responses about healthcare providers. A hospital with 4.8 stars across 2,000 verified reviews will appear in AI recommendations for "best orthopedic surgeon in [city]" more reliably than one with 3.9 stars and 200 reviews. This isn't new -- but the weight AI models place on these signals is higher than traditional search.
Third-party citations and media coverage
When reputable medical publications, peer-reviewed journals, or major news outlets cite your institution or drug, AI models pick that up. A study published in JAMA that references your clinical outcomes is worth more to your AI visibility than ten press releases on your own website.
Accurate, current content on your own domain
AI crawlers visit your site. If your content is outdated, thin, or contradicts what third-party sources say about you, you lose credibility in the model's synthesis. Regular content audits -- especially for clinical programs, drug approvals, and specialist capabilities -- are not optional anymore.
What to actually monitor
Healthcare and pharma brands need to track several distinct categories of AI-generated content:
Brand mentions in response to condition-based queries. "What are the best hospitals for [condition]?" "Which hospitals perform [procedure]?" These are the queries patients actually use. Are you appearing? What are AI models saying about you when you do appear?
Drug and product descriptions. For pharma, this is the compliance-critical category. What does ChatGPT say about your drug's mechanism of action, approved indications, side effects, and efficacy compared to alternatives? Is it accurate? Is it current?
Competitor positioning. Which competitors appear when you don't? What are AI models saying about them that they're not saying about you? This is where content gap analysis becomes valuable -- not just knowing you're absent, but understanding why.
Sentiment and framing. Even when you appear, how are you described? "A regional hospital with a strong cardiac program" is very different from "a general hospital that also offers cardiac services." These framings shape patient decisions.
Source attribution. Which pages on your site are AI models actually citing? Which third-party sources are they pulling from? This tells you where to invest -- and what to fix.
Tools for AI brand monitoring in healthcare
The market for AI visibility monitoring has grown quickly, but not all tools are equally suited to healthcare and pharma use cases. Here's how the main options compare.
| Tool | AI engines monitored | Content gap analysis | Compliance-relevant features | Best for |
|---|---|---|---|---|
| Promptwatch | 10+ (ChatGPT, Perplexity, Gemini, Claude, etc.) | Yes -- Answer Gap Analysis | Crawler logs, page-level citation tracking, source analysis | Full-cycle monitoring + optimization |
| Profound | 9+ AI engines | Limited | Strong enterprise reporting | Enterprise monitoring |
| Otterly.AI | ChatGPT, Perplexity, Google AI Overviews | No | Basic monitoring dashboard | Simple tracking |
| Peec AI | ChatGPT, Perplexity, Claude | No | Monitoring only | Small teams |
| ScrunchAI | Multiple LLMs | No | Monitoring + basic reporting | Mid-market brands |
| LLM Pulse | ChatGPT, Perplexity, and more | No | Basic visibility tracking | Budget monitoring |
For healthcare and pharma specifically, the compliance dimension pushes the requirements beyond what basic monitoring tools can handle. You need to know not just whether you're being mentioned, but what is being said, which sources are being cited, and whether any AI-generated content about your products raises regulatory flags.
Promptwatch is the platform I'd point healthcare teams toward first, because it closes the loop between monitoring and action. It shows you which prompts competitors rank for that you don't, lets you see exactly which pages AI models are citing, and includes a built-in content generation tool for creating material that's designed to get cited. For pharma teams worried about AI models surfacing inaccurate drug information, the source analysis feature is particularly useful -- you can see what AI models are pulling from and prioritize correcting or displacing those sources.
For teams that want a dedicated enterprise-grade option with strong reporting, Profound is worth evaluating.
Profound
For simpler use cases -- a regional hospital system that just wants to know if it's appearing in AI responses for key conditions -- Otterly.AI or Peec AI may be sufficient starting points.
Otterly.AI

Building a compliance-aware monitoring workflow
Here's a practical workflow for healthcare and pharma teams that takes compliance seriously.
Step 1: Define your prompt universe
Start with the queries that matter most to your business. For a hospital system, this might include:
- Condition-based queries ("best hospital for [condition] in [region]")
- Procedure queries ("where to get [procedure]")
- Comparative queries ("[your hospital] vs [competitor hospital]")
- Reputation queries ("is [your hospital] good?")
For pharma, add:
- Drug information queries ("how does [drug name] work?")
- Comparison queries ("[drug name] vs [competitor drug]")
- Safety queries ("side effects of [drug name]")
- Indication queries ("what is [drug name] used for?")
The compliance-sensitive queries are the ones where inaccurate AI output creates regulatory risk. Flag these separately and monitor them more frequently.
Step 2: Run baseline monitoring across multiple AI engines
Don't just check ChatGPT. Patients and physicians use Perplexity, Google AI Overviews, Gemini, and increasingly Claude and Copilot. Each model has different source preferences and citation patterns. A drug description that's accurate in ChatGPT might be outdated in Perplexity if the models are pulling from different source sets.
Step 3: Document and categorize findings
For each monitored prompt, record:
- Whether your brand appears
- What is said about you
- Which sources are cited
- Whether any claims are inaccurate, outdated, or potentially off-label (for pharma)
- How you compare to competitors in the same response
This documentation matters for compliance purposes. If an AI model consistently surfaces off-label information about your drug, having a record of when you identified the issue and what steps you took to address it is relevant to your regulatory posture.
Step 4: Identify content gaps and create authoritative content
If AI models aren't citing your content, the most likely reason is that your content doesn't exist or isn't authoritative enough. This is where the monitoring workflow connects to content strategy.
For each gap you identify, ask: what would a patient or physician need to read to get accurate information about this topic? Then create that content, authored by named clinical experts, with proper schema markup, and published on your domain.
Promptwatch's Answer Gap Analysis makes this systematic -- it shows you the specific prompts where competitors appear and you don't, so you can prioritize content creation based on actual AI search demand rather than guessing.
Step 5: Track changes over time
AI models update their knowledge and source preferences continuously. A content piece you published last month might start getting cited next month -- or a previously accurate AI response might drift as the model incorporates new sources. Regular monitoring (weekly for high-priority prompts, monthly for the broader set) catches these changes before they become problems.
Trust signal optimization for healthcare brands
Beyond monitoring, there are specific actions that improve how AI models perceive and cite healthcare brands.
Structured data and schema markup. Use MedicalOrganization, Hospital, Physician, and Drug schema types. These give AI crawlers explicit, machine-readable signals about what your organization does and who your clinical staff are.
Claim your profiles on authoritative directories. Healthgrades, US News Health, Vitals, WebMD's provider directory -- these are sources AI models trust. Ensure your listings are accurate, complete, and regularly updated.
Publish clinical outcomes data. AI models weight clinical evidence heavily. If your hospital publishes outcomes data, survival rates, or complication rates, that content is more likely to be cited than marketing copy.
Respond to reviews. AI models pick up on review response patterns as a trust signal. A provider that responds thoughtfully to negative reviews signals accountability. One that ignores them signals the opposite.
Build external citations. Encourage your physicians to publish in peer-reviewed journals, contribute to medical education content, and participate in professional associations. Each external citation strengthens the signal that your organization is authoritative.
Monitor Reddit and patient forums. This one surprises some healthcare marketers, but AI models like Perplexity actively cite Reddit discussions and patient community forums. If patients are sharing negative experiences in r/AskDocs or condition-specific subreddits, those discussions may be shaping AI responses about your brand. Monitoring these channels -- which Promptwatch does as part of its Reddit tracking -- gives you early warning.
The regulatory horizon in 2026
The compliance environment for AI in healthcare is moving fast. The February 2026 consultation signals from aihealthcarecompliance.com indicate that regulators are beginning to treat AI systems with patient safety implications as high-impact, requiring more rigorous oversight.
For pharma specifically, the question of who is responsible when an AI model surfaces off-label drug information is unresolved. The FDA's current guidance on digital health and AI doesn't cleanly address the scenario where a third-party AI model -- not your own promotional materials -- makes an inaccurate claim about your product. But that regulatory gap is unlikely to persist. Proactive monitoring and documentation now is the sensible posture.
For hospital systems, the concern is slightly different: AI models that describe your capabilities inaccurately can redirect patients who need specialized care to the wrong facility. That's a patient safety issue that goes beyond marketing.
The brands that will be best positioned as regulations tighten are the ones that have already built systematic monitoring, documented their findings, and invested in authoritative content that gives AI models accurate information to cite.
Where to start
If you're a healthcare or pharma marketer reading this and you haven't started monitoring AI brand mentions yet, the first step is simple: run a handful of your most important queries through ChatGPT, Perplexity, and Google AI Overviews manually. See what comes back. Note whether you appear, what's said about you, and whether any of it is inaccurate.
That manual audit will tell you whether you have a problem. It won't tell you the full scope of it, and it won't scale. For systematic monitoring across multiple AI engines, with the ability to track changes over time and identify content gaps, you need a dedicated platform.
The compliance stakes in healthcare and pharma make this a higher-priority investment than it might be in other industries. An AI model that misrepresents your drug's approved uses or describes your hospital's capabilities inaccurately isn't just a marketing problem. It's a patient trust problem, and potentially a regulatory one.
Start monitoring. Document what you find. Fix the gaps. That's the loop.